An Antiviral Antiseptic Gargle & Nasal Spray May Help Healthy People Avoid Coronavirus Infection
April 1, 2020 7:47 pm
Introduction:
An antiviral antiseptic gargle & nasal spray may help healthy people avoid coronavirus infection. The use of an antiviral antiseptic is supported in the scientific medical literature, based upon coronaviruses similar to COVID-19, as a preventive addition to self-quarantine, social distancing, respiratory hygiene and Personal Protective Equipment (“PPE”). COVID-19 and Coronavirus (“CV”) refer to the same virus for this discussion.

An antiviral antiseptic intraoral gargle & intranasal spray may be an additional line of protection for a healthy, non-infected person if coronavirus gets past their own face mask and enters their own nose or mouth. Unfortunately it appears that one in five new coronavirus infections in the U.S. as of April 7 2020 appear in healthcare workers and civil servants trying to help infected patients. Clearly PPE is both unavailable and insufficient. And a vaccine will not be available for the general public soon enough. Therefore this webpage continues to assist medical and dental schools, hospitals and the U.S. military. The Chief of the US Army Dental Corps, the highest ranking dentist in the US Army, forwarded this document to the CDC for review on March 31 2020. He also forwarded it to the Tri-Service (Army, Navy & Air Force) Infection Prevention Control Team for discussion on April 16 2020. This is not a “treatment” nor cure for those who are already infected though it may help lower the infection risk they pose to others. The CDC offers no guidance on using antiviral antiseptics against COVID-19 because it is so new, therefore no claim is made.
It is easy and inexpensive ($20) to purchase everything that is needed online without a prescription. It is also easy to make at home. It appears 0.5% Povidone-Iodine (PVP-I) is the ideal antiviral antiseptic for use as an intraoral gargle and intranasal spray because it has proven toxicity to many other coronaviruses that are similar to COVID-19. Different brands of intraoral gargles and intranasal sprays containing PVP-I have been sold for decades around the world including Japan. They were not considered commercially successful in the U.S. because their use can stain teeth over time. More info on safety, how to purchase and how to make at home is provided below.
This webpage began as a scientific literature review written on March 21, 2020 as COVID-19 began killing a lot of people. It is being provided for the public good during a crisis. It is based upon the latest medical research, published or submitted scientific papers in academic journals, and thinking in prestigious medical and dental schools. Many academic references are provided below in the Supporting Science and Salient References section. Please forward this information to your primary care doctor, dentist or nurse for personal recommendations. The author has no financial interest in the information presented and is not selling any associated product nor service. Dr. Dorfman retired and closed his private practice in 2019.
Dr. Jeffrey Dorfman
1dentist, LLC
New York
https://www.linkedin.com/in/jeffreydorfman/

Photo Caption: This is NOT a drink. It contains 0.5% PVP-I diluted 20:1 in distilled water. Any thoroughly cleaned container that is over 20 ounces in size can be used.
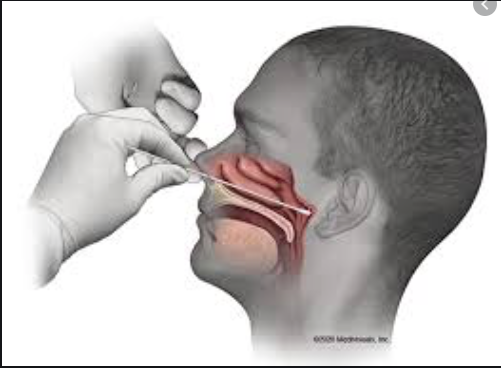
The route of entry of coronavirus into the body is thought to be first the nose, then the mouth and lastly the eyes. How the Coronavirus Attacks Your Body offers a clear explanation. That is why front line healthcare workers use cotton swabs in the back of the nose and throat to test to see who is infected.
The preventive concept presented here is to make the oral and nasal mucous membranes of a healthy person less hospitable to the initial viral inoculation. It is believed that the majority of COVID-19 infection in the lungs first attaches to these mucosal surfaces in the nose & throat and then replicates over several days before entering the lower respiratory tract. Direct inhalation into the lungs is considered much less common. It is also suggested, in Methods/Protocol for Hospitals below, that this antiviral antiseptic may help make those already infected with COVID-19 less infectious to others.
In normal times we need Randomized, double-blind Clinical Trials (“RCT”) to prove the efficacy of any drug. However we are not living in normal times. The government, FDA, CDC, & state governments are trying many logical ideas to offer immediate help in an attempt to save lives. Why not this antiviral antiseptic?
Coronavirus testing in the above photo is performed by healthcare personnel using a long Q-tip through the nose into the nasopharynx. This area is considered to be the primary area of infection. Another area of infection is through the mouth into the oropharynx. The oropharynx is the area in the back of the throat just below the end of the Q-tip seen in the photo. The nasopharynx and oropharynx join at about the height of the chin then provide a pathway to the lungs. An antiviral antiseptic in the nose and mouth may help prevent infection in these areas. It may also help reduce the likelihood of those already infected from infecting others.
Safety:
An “antiseptic” can be used in or on the body. A “disinfectant” can be used to clean inanimate surfaces like kitchen counters but not in, or on, the body. Oral and nasal antiseptic products containing PVP-I are available in the U.S. Mouthwashes containing PVP-I were thought to be commercially unsuccessful because frequent use can result in staining the teeth. However the safety profile of PVP-I has been well-established. Betadine® is a well known brand for external wound care antiseptic that contains 10% PVP-I. Generic 10% PVP-I works fine and is much less expensive.10% PVP-I is too high a concentration to use in the mouth and nose! Allergy is discussed further below in the Exclusion Criteria and Allergy section.
How to Make at Home: 0.5% PVP-I Mouthwash and Nasal Application with a Q-tip or Spray:
The Povidone-Iodine (PVP-I) mouthwash and nasal application is easy to make at home. This is NOT a drink. Consult with your doctor, dentist or nurse before use. This information is based upon the protocol offered in hyperlink #3 in the Supporting Science and Salient References section below.
Photo Caption: This is NOT a drink. Buy PVP-I 10% liquid at a local pharmacy or online: Generic or Betadine®.
This is NOT a drink! Instead it should be gargled in the back of the throat for 30 seconds then spit out. Do not rinse with water afterwards. Avoid eating or drinking anything afterwards for 30-60 minutes. We need 0.5% PVP-I in distilled* water (20:1 dilution of generic PVP-I, 10%). Diluting a 10% concentration liquid by 20 equals a concentration of 0.5%. The diluted 0.5% concentration is needed for both the gargle and nasal application.
It is easiest to start with an 8 ounce bottle of PVP-1, 10% concentration. PVP-I may be purchased without a prescription at a local pharmacy or online. A 20:1 distilled water to PVP-I dilution means 19 ounces of distilled water should be added to one ounce of 10% PVP-I in any thoroughly cleaned container that is somewhat larger than 20 ounces. The result is 1 ounce of PVP-I in 20 ounces of total liquid (20:1).
Pour 1/3 of an ounce (2 teaspoons) of the mixture into any glass or cup. Gargle then spit as described above. A shot glass is easy to measure by sight because 1/3 ounce is about 1/4 the inside height. The exact volume is not critical but try to be close.
*Tap water may be used for the gargle instead of distilled water but it cannot be used in the nose. Only distilled water or boiled (and cooled) tap water may be used in the nose otherwise a serious sinus infection can occur. Iodine in the PVP-I turns the distilled water brown.

Photo caption: Nasal spray atomizer. Buy at a local pharmacy or online. More online options for a vertical not horizontal sprayer.
The nasal application may be made in one of two ways:
1, Dip both ends of a Q-tip into the mouthwash before it is gargled and then apply in a circle in both nostrils.
2. Pour some of the mouthwash into a nasal spray atomizer bottle that you can purchase. Then spray twice in each nostril. The nasal spray atomizer will provide better coverage deeper inside the nose and deeper into the nasopharynx. Most standard nasal spray atomizers dispense 0.1ml in each spray. Verify if possible.
Purchase only three different items. Rx not needed. Cost about $20.
1) PVP-I, 10% solution (Povidone-Iodine, 10% solution) one 8 ounce bottle from a local pharmacy or online. Rx not needed. Betadine® is a well known brand for external wound care antiseptic that contains 10% PVP-I. A generic PVP-I, 10% is fine and less expensive. Buy at a local pharmacy or online: Generic or Betadine®.
2) Nasal spray atomizer bottle (that sprays vertically into the nose and not horizontally). Buy at a local pharmacy or online. More online options. Buy one per person.
3) Distilled water in gallon containers from any local store or online. An 8 ounce bottle of PVP-I will require 152 ounces of distilled water for mixing. This is slightly more than one gallon (128 ounces). Therefore buy two gallons of distilled water.
Dosage:
It is suggested to reapply the mouthwash and nasal application every three hours up to four times a day. Healthcare personnel and others at risk will need this frequency of application. People who are mostly staying at home can use it less frequently per day depending upon their activities and risk of exposure to coronavirus. Some minimal frequency of use may still be worthwhile even for those self-quarantined.
One 8 ounce bottle of PVP-I, 10% combined with 19 ounces of distilled water yields 20 ounces of the desired 0.5% concentration PVP-I. Using 0.3 ounce per dose (2 teaspoons) means each 20 ounce bottle yields 60 doses of the intraoral gargle. Therefore one 8 ounce bottle plus less-than two gallons of distilled water should yield close to 500 doses. One 1-ounce nasal spray atomizer bottle could yield 2-5 weeks of coverage if used between two to four times per day.
Exclusion Criteria and Allergy:
There is no recommendation to use this mouthwash in children, during pregnancy or in those who are medically ill. Everyone considering the use of this mouthwash should forward this information to their physician or dentist for personal recommendations. Please read the disclaimer at the bottom of this page.
Iodine itself is not considered an antigen, i.e. it does not cause an allergic response. Iodine is an essential trace mineral that is needed in our diet. Iodine deficiency is the leading cause of intellectual and developmental disabilities affecting two billion people in the world. That is why most table salt contains iodine and is referred to as “iodized salt.”
The University of California San Francisco states that no evidence exists to suggest that the iodine content of seafood is related either to seafood intolerance or seafood allergy. Instead these reactions are frequently associated with transmission of infection in uncooked food, the presence of toxins within the seafood or a response to fish muscle protein.
A history of allergy to PVP-I or its relevant excipients, autoimmune thyroid disease or current radio-active iodine treatment are reasons not to use PVP-I. Betadine® brand for external wound care contains 10% PVP-I. However it also includes other trace chemicals like Pareth 25-9 and Sodium Hydroxide that might cause a few allergic reactions. Therefore a bottle of generic PVP-I, 10% is preferred. Povidone can also cause a few allergic reactions. A severe allergic reaction can result in death. However the risk of an allergic reaction in the general population is low compared to the lethality of COVID-19.
Antiseptic alternatives to 0.5% PVP-I that appear in current research literature include: 1.5% Hydrogen Peroxide, 0.12% Chlorhexidine, 0.1% Sodium Hypochlorite and sterile hypertonic saline solution. However 0.5% PVP-I appears to be the first choice against COVID-19 because of its proven toxicity against other coronaviruses. This author is simply interested in using the best antiviral antiseptic gargle & nasal spray against COVID-19.
Supporting Science:
I had two questions: 1) Did an antiviral antiseptic exist that would kill COVID-19, and then, 2) could it provide antiviral activity in vivo in the mouth and nose? It appears the answer is yes to both questions. An intraoral & intranasal antiviral antiseptic may help reduce transmission of COVID-19. It is not a treatment for those who are already infected.
It appears Povidone-Iodine (PVP-I) is the ideal antiseptic. The first hyperlink below contains my current literature review on the ideal antiviral antiseptic and supports CHX. I initially favored Chlorhexidine (CHX) as the preferred antiseptic because of its inherent substantivity (“stickiness”) to mucosal tissue; CHX lasts four times longer in the mouth than PVP-I. CHX also helps prevent Ventilator-Associated Pneumonia (VAP) in studies conducted before the existence of COVID-19. Many COVID-19 patients die from pneumonia. The second hyperlink, provided by Dr. Stephen Challacombe, discusses the inactivation of human viruses and supports PVP-I. The third hyperlink, by Dr. Stephen Challacombe et al, suggests a specific method and protocol for immediate use of PVP-I as an adjunct to currently recommended social distancing and PPE.
I was convinced to support PVP-I over CHX after discussions with Penn Dental School Dean Mark Wolff, Dr. Victor Badner at Jacobi Hospital and NYU Dental School Oral Medicine professor, Dr. Ross Kerr. Dr. Kerr shared Dr. Stephen Challacombe’s reference and his entire unpublished paper with me on March 29. Ultimately, PVP-I is more virucidal than CHX.
Salient References. The underlined portions are hyperlinks:
- Chlorhexidine (CHX) vs COVID-19 Literature Review – my initial review includes a lot of other academic references.
- Inactivation of Human Viruses by Povidone-Iodine in Comparison with Other Antiseptics – Derm 1997
This reference from Dr. Stephen Challacombe was provided via personal email from Dr. Ross Kerr on March 29.
- The Use of Povidone Iodine Nasal Spray and Mouthwash During the Current COVID-19 Pandemic
The entire paper by Dr. Stephen Challacombe et al was provided via personal email from Dr. Ross Kerr on March 29. It awaits publication however it can be downloaded from this page. - In Vitro Bactericidal and Virucidal Efficacy of Povidone-Iodine Gargle/Mouthwash Against Respiratory and Oral Tract Pathogens – Infect Dis Ther 2018.
- Infectious Disease Management and Control with Povidone Iodine – Infect Dis Ther 2019.
- A pilot, open labelled, randomised controlled trial of hypertonic saline nasal irrigation and gargling (HSNIG) for the common cold – Nature 2019. Gargling and irrigation are thought to facilitate viral shedding.
- Topical povidone iodine inhibits bacterial growth in the oral cavity of patients on mechanical ventilation: a randomized controlled study – Feb 2020.
- Comparison of 5% povidone-iodine solution against 1% povidone-iodine solution in preoperative cataract surgery antisepsis: a prospective randomised double blind study – Br J Ophthalmol. 2003. PVP-I used in the eye.
- Efficacy of povidone-iodine vaginal suppositories in the treatment of bacterial vaginosis – Dermatology 2002. PVP-I used in the vagina.
- Hospitalization Rates and Characteristics of Patients Hospitalized with Laboratory-Confirmed Coronavirus Disease 2019 — COVID-NET, 14 States, March 1–30, 2020 – CDC 2020. “Respiratory hygiene” is mentioned three times in this paper.
- Potential Role of Oral Rinses Targeting the Viral Lipid Envelope in SARS-CoV-2 Infection. May 14 2020. Oxford University Press on behalf of the American Physiological Society. “We highlight that already published research on other enveloped viruses, including coronaviruses, directly support the idea that oral rinsing should be considered as a potential way to reduce transmission of SARS-CoV-2.”
- COVID-19: Povidone-Iodine Intranasal Prophylaxis in Front-line Healthcare Personnel and Inpatients (PIIPPI). April 28, 2020. Clinical trial posted for 250 participants at the University of Kentucky.
- Halodine® Nasal and Oral Antiseptics Show Rapid Antiviral Activity Against SARS-CoV-2 (COVID-19). May 13, 2020. A new commercial product.
- The Ocular Application of Povidone-Iodine. Community Eye Health 2003. PVP-I has a long history of use in the eye. Therefore it is an antiviral antiseptic that can be used in the mouth, nose and eyes. This is one of many articles on this topic.
- Consideration of povidone-iodine as a public health intervention for COVID-19: Utilization as “Personal Protective Equipment” for frontline providers exposed in high-risk head and neck and skull base oncology care. June 2020. Oral Oncology.
This information is a summary of many emails particularly between myself; Dr. A. Ross Kerr, Clinical Professor of Oral Medicine at New York University (NYU) College of Dentistry; Dr. Stephen J. Challacombe, Professor of Oral Medicine at King’s College in London and COL David Ferguson, DDS, MS, Office of the Chief, U.S. Army Dental Corps. NYU Dental School Dean Dr. Charles Bertolami has been very supportive of this effort. There are a lot of others to personally thank including Neal Naito, M.D. who wrote the New York Times article, “Gargling for Coronavirus? What Science Can Tell Us,” linked below and the many insightful commenters who discussed his article.
Methods/Protocol for Hospitals:
Dr. Stephen Challacombe (“SC”) is a world-renowned research scientist and Professor of Oral Medicine at King’s College in London. The third hyperlink above will allow you to download his entire seven page paper. Methods/Protocol and method of application are on pages 3-4. Ideally, the paper suggests starting this protocol now for all patients and healthcare personnel.
SC recommends applying 0.5% PVP-I to the oral, oropharyngeal and nasopharyngeal mucosa of patients with presumed/confirmed COVID-19 and the healthcare personnel in close contact with them. He recommends it for all procedures “in or around the mouth and nose or procedures that transit those areas.” The clinician may decide to exclude some very sick patients due to concerns of aspiration etc.
Additionally, SC recommends this 0.5% application of PVP-I to all patients, not just those with suspected/confirmed COVID-19 infection. This would then suggest that all healthcare personnel be similarly treated. Therefore all patients and healthcare personnel, COVID-19 negative and positive, should receive this protocol. This is concept is not limited to dental patients. This concept should therefore also be considered for the general public. However I leave this recommendation to the CDC.
This Method/Protocol may be considered to augment prevention of disease transmission rather than treatment of the disease. Infected patients exhibit a very high viral load in saliva (mouth & oropharynx) and an even higher viral load in the nose (nasopharynx). PVP-I may lower the viral load in the mouth and nose in these infected patients and therefore may reduce their risk of infecting healthcare personnel who are already using all available PPE. Furthermore, it may be considered prevention rather than treatment when this protocol is used on all non-infected healthcare personnel because the route of viral entry is the mouth, nose and eyes. The preventive concept is to make the oral and nasal mucous membranes of a healthy person less hospitable to the initial viral inoculation.
Randomized, Double-Blind Clinical Trials (“RCT”) Needed:
A Randomized, double-blind Clinical Trial (“RCT”) could offer a large group of healthy people a mouthwash and nasal spray containing one of the following:
1) PVP-I, 0.5%
2) Chlorhexidine, 0.12%
3) H2O2, 1.5%
4) Sodium Hypochlorite, 0.1% [Click for more info]
5) Hypertonic saline
6) H2O as a control
Then simply measure over time the incidence of these five groups contracting COVID-19. The military may offer ideal relatively-closed environments for testing.
This RCT is relatively simple & inexpensive to perform. It will also offer real-time info. It is not necessary to work with live virus. Therefore a Biosafety Level 3 (“BSL3”) facility is not needed. However Institutional Review Board (“IRB”) approvals and enrollment & administrative staff are needed.
The NY Times published my Comments along with many others of considerable value on April 1 2020.

Disclaimer:
This info is not intended to offer advice. The preventive benefit of an antiviral antiseptic against COVID-19 has not been established by the CDC. Instead it is suggested for consideration by the CDC as an adjunct to self-quarantine, social distancing, respiratory hygiene and Personal Protective Equipment (“PPE”). Please forward this information to your primary care doctor, dentist or nurse for personal recommendations. The author has no financial interest in the information presented and is not selling any associated product nor service. Dr. Dorfman retired and closed his private practice in 2019.
Dr. Jeffrey Dorfman
1dentist, LLC
New York
https://www.linkedin.com/in/jeffreydorfman/
Categorised in: Uncategorized
This post was written by Dr. Jeffrey Dorfman